What is the purpose of the Transform Study?
Efforts to reduce heart attack, stroke and other cardiovascular events by managing risk factors have met with limited success. TRANSFORM is designed to find out if a Personalized Care Strategy based on treating an individual’s actual burden of atherosclerotic plaque is more effective.
What is the purpose of Coronary Computed Tomography Angiography (CCTA)?
Coronary Computed Tomography Angiography (CCTA) is an X-ray which produces 3-dimensional images of fatty build up in the arteries supplying blood to the heart (coronary plaque). The Cleerly staging system analyzes the images to assess each individual’s risk of cardiovascular events such as heart attack, stroke, heart failure, peripheral artery disease (blockages in blood flow to the legs) and procedures to improve blood flow.
Who can participate in the Study?
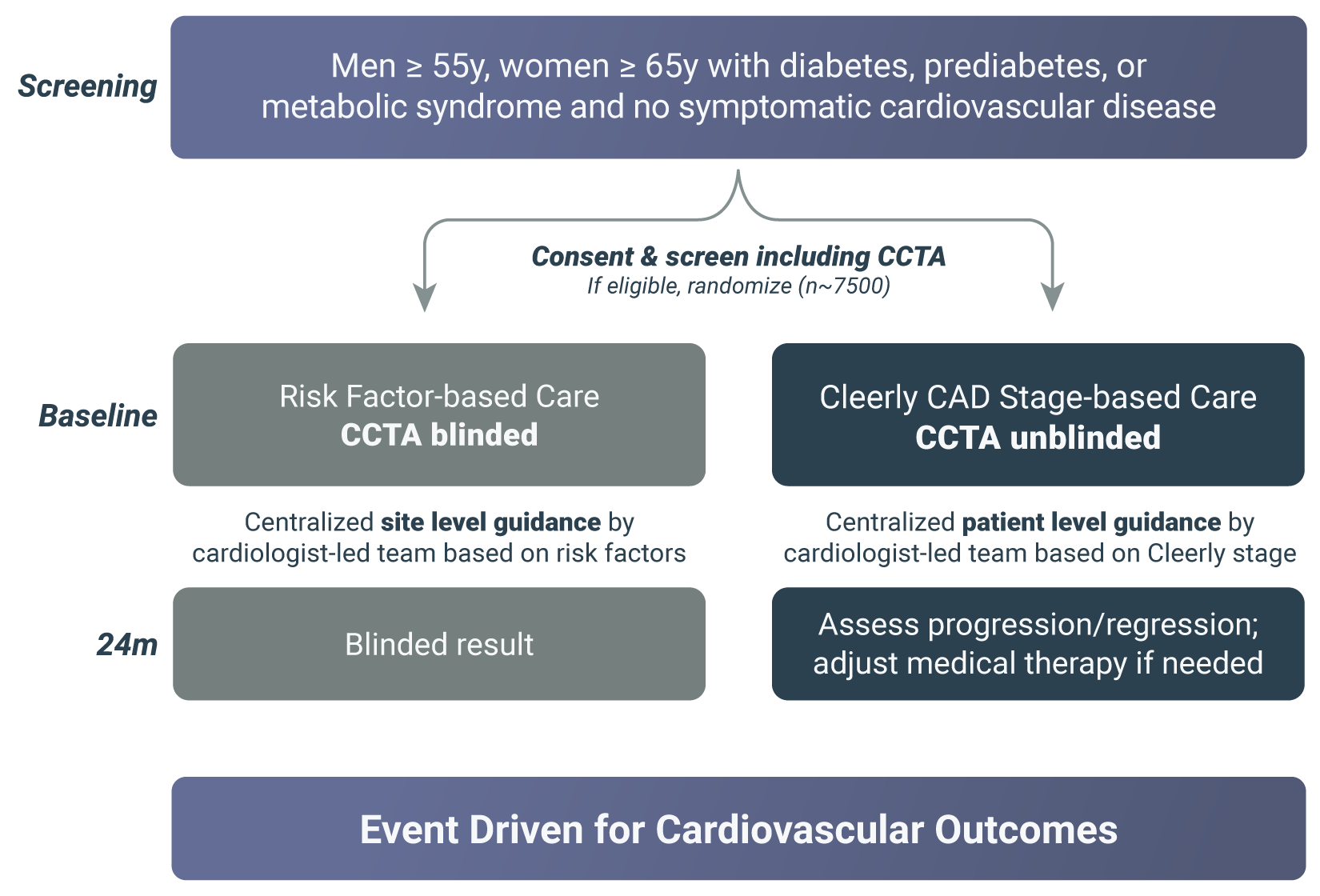
Men >55y, women >65y with diabetes, prediabetes or metabolic syndrome and no symptomatic cardiovascular disease can participate in the study.
How does this trial work?
Candidate participants will have a CCTA to see if the study is right for them If it is determined that participation in the study is possible, participants will be assigned at random (like flipping a coin) to the personalized care group or usual care group Participants in the usual care group will receive health care from their usual care provider Participants in the personalized care group will receive health care from a remote cardiologist-lead team which will individualize treatment based on the Cleerly CAD staging results of their CCTA Both groups will report healthcare events such as heart attack, stroke, heart failure or procedures to improve blood flow, and occurrence of these events in the 2 groups will be compared.
How many people and countries are participating in the study?
TRANSFORM will enroll approximately 7500 people in the United States.
How long will it take to finish the study?
The total study duration is expected to be 5 years.
Where is the Transform trial being conducted?
The Transform trial is being conducted at research sites in the United States. For the Usual care group, the primary care physician will provide medical consultation and drug prescription and monitor the drug's safety. The virtual cardiologist’s team will provide care for the Personalized care group, including consultation, drug prescription, and monitoring via a computer platform. Heartbeat Health (HBH) takes responsibility for it. HBH(Link is needed) is a technology-enabled virtual cardiology provider leveraging real-time clinical data & device connectivity to diagnose and deliver proactive heart care.
Are there any substudies that are a part of the Transform trial?
We intend to study three themes through this project. 1. TRANSFORM-Outcomes - A study on whether personalized care reduces cardiovascular events compared to usual care. 2. TRANSFORM-Plaque - Whether this tailored approach can improve the amount and aspect of plaque on f/u CCTA. 3. TRANSFORM-Classify - Determining whether the diagnostic technique based on CCTA has an advantage over existing techniques.